Fruit Flies Experiment
The fruit fly is also known as Drosophila. This organism does not have a large number of chromosomes. Since the fruit fly is small and has simple living necessities, it is an great organism for learning about genetics. If the fruit fly is going through their life cycle in room temperature, it can take about 12 days to complete. Typically, fruit flies take around 2 weeks to fully develop into an adult. The life cycle of the fruit fly consists of four stages: egg, larva, pupa, then adult. A female fruit fly can lay about five eggs at a time, which is another reason fruit flies are great organisms to study genetics (they produce a lot of offspring). The egg is oval shape and small. It takes about a day for the egg to hatch into larva. In the larval stage, the fruit fly larva is wormlike, constantly eats, can move through a medium when it eats, and undergoes two molting periods. The larva tends to be difficult to see. However, a larva has a mouth that is black that is easy to see. When the larva is ready for the next stage, it wiggles away from their medium to form a puparium. The egg and larval stage typically takes around eight days. The next stage is the pupal stage. This stage is when the fruit fly undergoes metamorphosis. When the fruit fly is ready to emerge from the puparium, the pupal case becomes hard and dark. A day before the fruit fly is ready to go into the next stage, the eyes, wings, and legs will be visible in the puparium. The fruit fly typically spends six days in the pupal stage. When the fruit fly completes metamorphosis, it starts the adult stage. The fruit fly can spend up to several weeks in this stage. When the fruit fly emerges from the puparium, the fruit fly’s color may be light. The fruit fly will also have an abdomen that is long and the wings will not expand. This appearance my last a few hours. The fruit fly with then get dark, round, and spread its’ wings. Female fruit flies can mate after 8-12 hours after they begin their adult stage. After a couple days in the adult stage, females can begin to lay eggs. The female fruit fly can contain sperm in their systems to reproduce in the future.
The sex of the fruit fly can be determined by the abdomen and forearms. Male fruit flies have sex combs (black comb-like spikes) on their forearms, rounder abdomens with 5 segments. Female fruit flies tend to have longer abdomens with seven segments.
Fruit flies also have mutations that make them look different from each other. They can differ in eye and body color. Wild type fruit flies (also known as tan body fruit flies) are the non-mutated fruit flies. Short winged fruit flies do not fly and possess a recessive trait (inherited by both parents). Curly winged fruit flies possess a dominant trait (can be inherited by only one parent). This trait does not allow the fruit fly to fly. However, the fruit fly will have the ability to jump around from place to place. Yellow fruit flies have a recessive mutation. These fruit flies do not have dark pigmentation. Ebony fruit flies also have a recessive mutation. These fruit flies possess an all-over dark body. Mutations can also cause different color eyes for fruit flies. Fruit flies with white eyes or orange eyes have recessive mutations. White eyed fruit flies have mutations on their sex chromosomes. The eye mutations are caused when the red eye chromosome is damaged. Fruit flies may also be eyeless. Therefore, eyeless fruit flies are unable to see. This mutation is also recessive. Antennapedia is when a fruit fly has a dominant mutation that causes it to possess an extra pair of legs where the antennas are suppose to be.
In this experiment, the ebony mutation will be examined. The ebony fruit flies will be crossed with the typical wild type fruit flies. The ebony mutation is on chromosome 3 of the fruit fly. This trait is autosomal recessive.
The sex of the fruit fly can be determined by the abdomen and forearms. Male fruit flies have sex combs (black comb-like spikes) on their forearms, rounder abdomens with 5 segments. Female fruit flies tend to have longer abdomens with seven segments.
Fruit flies also have mutations that make them look different from each other. They can differ in eye and body color. Wild type fruit flies (also known as tan body fruit flies) are the non-mutated fruit flies. Short winged fruit flies do not fly and possess a recessive trait (inherited by both parents). Curly winged fruit flies possess a dominant trait (can be inherited by only one parent). This trait does not allow the fruit fly to fly. However, the fruit fly will have the ability to jump around from place to place. Yellow fruit flies have a recessive mutation. These fruit flies do not have dark pigmentation. Ebony fruit flies also have a recessive mutation. These fruit flies possess an all-over dark body. Mutations can also cause different color eyes for fruit flies. Fruit flies with white eyes or orange eyes have recessive mutations. White eyed fruit flies have mutations on their sex chromosomes. The eye mutations are caused when the red eye chromosome is damaged. Fruit flies may also be eyeless. Therefore, eyeless fruit flies are unable to see. This mutation is also recessive. Antennapedia is when a fruit fly has a dominant mutation that causes it to possess an extra pair of legs where the antennas are suppose to be.
In this experiment, the ebony mutation will be examined. The ebony fruit flies will be crossed with the typical wild type fruit flies. The ebony mutation is on chromosome 3 of the fruit fly. This trait is autosomal recessive.
Methods
Homozygous dominant male tan body fruit flies will be crossed with homozygous dominant female ebony body fruit flies in this experiment. Homozygous dominant female tan body fruit flies will also be crossed with homozygous dominant male ebony fruit flies. To start this experiment, a vial must be created for the fruit flies to live in. To create a living environment for the fruit flies, white flakes that turn into a blue gel-like substance when wet will be needed along with a vial (with a foam lid) and yeast. To create this vial, 2 inches worth of white flakes must be placed in the vial. An equal amount of water must also be added to create a blue gel-like substance. After the gel-like substance is created, a small amount of yeast should be sprinkled on top. This completes the living environment for the fruit flies. Two vials will be needed in the beginning.
True breed wild and ebony type fruit flies, fly nap, and swabs will then be needed for the experiment. The true breed fruit flies should be in 2 different vials. One vial should contain true breed wild type fruit flies while the other vial should contain true breed ebony type fruit flies. Two swabs should be soaked in fly nap until it is damp. After there are two damp fly nap swabs, the swabs should be placed into the true breed vials. It is important to make sure the fly nap swabs do not touch the vials (this may cause the fruit flies to permanently sleep). It is also important not to remove the lid to prevent to fruit flies from escaping. When the fruit flies are sleeping, it is safe to remove the lid and pour out fruit flies to sex. About 3-5 fruit flies of each gender and trait should be counted and placed into 2 different vials. One vial will contain 3-5 homozygous dominant male tan body fruit flies and 3-5 homozygous dominant female ebony fruit flies. The second vial should contain 3-5 homozygous dominant female tan fruit flies and 3-5 homozygous dominant male ebony fruit flies. The vials should laying down so that the fruit flies do not fall into the gel-like substance. After the fruit flies are placed into the vials, the lid should be replaced. These vials are created for the fruit flies to mate. Each vial should be labeled with the correct cross. The vials should be placed in an incubator to maintain a constant temperature for the fruit flies.
Once the fruit flies wake up from the fly nap, they should begin to mate. When larvae is present in the vial, the parent fruit flies should be accounted for and removed. This will help differentiate the parents from the offspring (F1 generation). Once the fruit flies have reached the adult stage, they should be put to sleep and counted and recorded by their sex and phenotype. This count is the F1 generation. The F1 generation should be counted for two weeks. A new vial must then be created. After the fruit flies are counted, they should be placed into the new vial. Only place the F1 generation in the new vile during the first week of counting. This will allow the F1 generation to breed in a new vile. When larvae is present in the second vial, the F1 parents should be accounted for and removed. Once the F2 generation begins to hatch, the fruit flies should be counted and recorded based on their sex and phenotypes. This generation should be counted for two weeks. After two weeks of counting the F2 generation, the experiment is done.
Since the experiment started with true breeds, the offspring of the true breeds should be heterozygous dominant. The expected phenotypes for both crosses should be 100% tan body heterozygous. This is indicated in the Punnett Squares below. The expected phenotypes for the F2 generation should be 25% homozygous dominant, 50% heterozygous dominant, and 25% homozygous recessive. Therefore, 75% of the offspring should have tan bodies while only 25% of the offspring should have ebony bodies. 50% of the offspring will be carriers for the recessive trait. This is indicated by the Punnett Squares on the next page.
True breed wild and ebony type fruit flies, fly nap, and swabs will then be needed for the experiment. The true breed fruit flies should be in 2 different vials. One vial should contain true breed wild type fruit flies while the other vial should contain true breed ebony type fruit flies. Two swabs should be soaked in fly nap until it is damp. After there are two damp fly nap swabs, the swabs should be placed into the true breed vials. It is important to make sure the fly nap swabs do not touch the vials (this may cause the fruit flies to permanently sleep). It is also important not to remove the lid to prevent to fruit flies from escaping. When the fruit flies are sleeping, it is safe to remove the lid and pour out fruit flies to sex. About 3-5 fruit flies of each gender and trait should be counted and placed into 2 different vials. One vial will contain 3-5 homozygous dominant male tan body fruit flies and 3-5 homozygous dominant female ebony fruit flies. The second vial should contain 3-5 homozygous dominant female tan fruit flies and 3-5 homozygous dominant male ebony fruit flies. The vials should laying down so that the fruit flies do not fall into the gel-like substance. After the fruit flies are placed into the vials, the lid should be replaced. These vials are created for the fruit flies to mate. Each vial should be labeled with the correct cross. The vials should be placed in an incubator to maintain a constant temperature for the fruit flies.
Once the fruit flies wake up from the fly nap, they should begin to mate. When larvae is present in the vial, the parent fruit flies should be accounted for and removed. This will help differentiate the parents from the offspring (F1 generation). Once the fruit flies have reached the adult stage, they should be put to sleep and counted and recorded by their sex and phenotype. This count is the F1 generation. The F1 generation should be counted for two weeks. A new vial must then be created. After the fruit flies are counted, they should be placed into the new vial. Only place the F1 generation in the new vile during the first week of counting. This will allow the F1 generation to breed in a new vile. When larvae is present in the second vial, the F1 parents should be accounted for and removed. Once the F2 generation begins to hatch, the fruit flies should be counted and recorded based on their sex and phenotypes. This generation should be counted for two weeks. After two weeks of counting the F2 generation, the experiment is done.
Since the experiment started with true breeds, the offspring of the true breeds should be heterozygous dominant. The expected phenotypes for both crosses should be 100% tan body heterozygous. This is indicated in the Punnett Squares below. The expected phenotypes for the F2 generation should be 25% homozygous dominant, 50% heterozygous dominant, and 25% homozygous recessive. Therefore, 75% of the offspring should have tan bodies while only 25% of the offspring should have ebony bodies. 50% of the offspring will be carriers for the recessive trait. This is indicated by the Punnett Squares on the next page.
Results
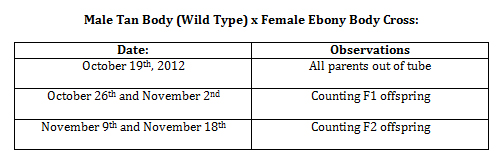
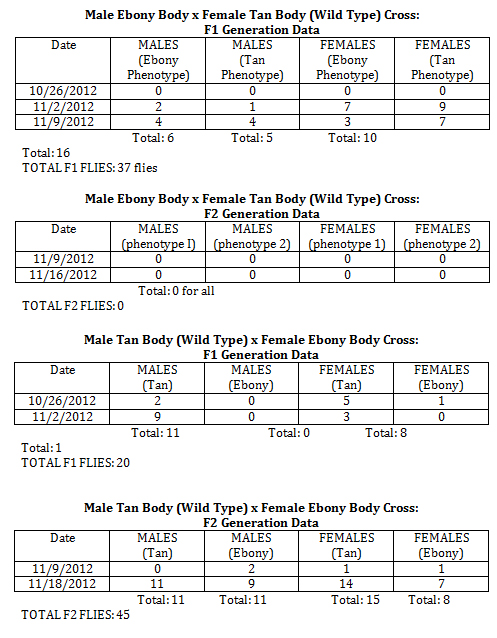
Observations and notes made during the experiment are in the tables below.
Phenotypes for all crosses can be seen below.
There is a significant difference between observed and expected values in the Chi Square Analysis in both crosses. The Chi-square Analysis for the two crosses can be seen in the data below.
CHI-SQUARE ANALYSIS:
Male Ebony Body x Female Tan Body (Wild Type) Cross: (4 Phenotypes, F1 Generation)
Number of Ebony Males: 6
Number of Ebony Females: 10
Number of Tan Males: 5
Number of Tan Females: 16
Total Number of Flies: 37
4 Integers = 37/4 = 9.25 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Males: X 2 = (6-9.25) 2 /9.25 = 1.142
Ebony Females: X 2 = (10-9.25) 2 /9.25 = 0.061
Tan Males: X 2 = (5-9.25) 2 /9.25 = 1.953
Tan Females: X 2 = (16-9.25) 2 /9.25 = 4.926
Total Chi Square : 1.142 + 0.061 + 1.953 + 4.926 = 8.082
Degrees of Freedom: 4-1=3
Probability (p-value) : 0.05 > p > 0.01
There is a significant difference between observed and expected values.
Male Ebony Body x Female Tan Body (Wild Type) Cross: (2 Phenotypes, F1 Generation)
Number of Ebony Males: 6
Number of Ebony Females: 10
Number of Tan Males: 5
Number of Tan Females: 16
Total Number of Flies: 37
2 Integers = 37/2 = 18.5 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Flies: X 2 = (16-18.5) 2 /18.5 = 0.338
Tan Flies: X 2 = (21-18.5) 2 /18.5 = 0.338
Total Chi Square : 0.338+0.338 = 0.676
Degrees of Freedom: 2-1 = 1
Probability Value (p-value) : 0.50 < p < 0.30
This p-value reflects that there is no significant difference between our observed values and the expected values in this experiment.
Male Tan Body (Wild Type) x Female Ebony Body Cross: (F1 Generation)
Number of Ebony Males: 0
Number of Ebony Females: 1
Number of Tan Males: 11
Number of Tan Females: 8
Total Number of Flies: 20
2 Integers = 20/2 = 10 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Tan Males: X 2 = (11-10) 2 /10 = 0.10
Tan Females: X 2 = (8-10) 2 /10 = 0.40
Ebony data : 0
Total Chi Square : 0.10+0.40 = 0.50
Degrees of Freedom: 2-1 = 1
Probability (p-value) : 0.50 < p < 0.30
This p-value reflects that there is no significant difference between our observed values and the expected values in this experiment.
Male Tan Body (Wild Type) x Female Ebony Body Cross: (F2 Generation)
Number of Ebony Males: 11
Number of Ebony Females: 8
Number of Tan Males: 11
Number of Tan Females: 15
Total Number of Flies: 45
Expected Numbers:
33.75: Tan flies
11.25: Ebony flies
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Flies: X 2 = (19-11.25) 2 /11.25 = 4.3
Tan Flies: X 2 = (26-33.75) 2 /33.75 = 1.77
Total Chi Square : 1.77+4.3 = 6.07
Degrees of Freedom: 2-1 = 1
Probability Value (p-value) : 0.05< p < 0.01
There is a significant difference between observed and expected values.
CHI-SQUARE ANALYSIS:
Male Ebony Body x Female Tan Body (Wild Type) Cross: (4 Phenotypes, F1 Generation)
Number of Ebony Males: 6
Number of Ebony Females: 10
Number of Tan Males: 5
Number of Tan Females: 16
Total Number of Flies: 37
4 Integers = 37/4 = 9.25 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Males: X 2 = (6-9.25) 2 /9.25 = 1.142
Ebony Females: X 2 = (10-9.25) 2 /9.25 = 0.061
Tan Males: X 2 = (5-9.25) 2 /9.25 = 1.953
Tan Females: X 2 = (16-9.25) 2 /9.25 = 4.926
Total Chi Square : 1.142 + 0.061 + 1.953 + 4.926 = 8.082
Degrees of Freedom: 4-1=3
Probability (p-value) : 0.05 > p > 0.01
There is a significant difference between observed and expected values.
Male Ebony Body x Female Tan Body (Wild Type) Cross: (2 Phenotypes, F1 Generation)
Number of Ebony Males: 6
Number of Ebony Females: 10
Number of Tan Males: 5
Number of Tan Females: 16
Total Number of Flies: 37
2 Integers = 37/2 = 18.5 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Flies: X 2 = (16-18.5) 2 /18.5 = 0.338
Tan Flies: X 2 = (21-18.5) 2 /18.5 = 0.338
Total Chi Square : 0.338+0.338 = 0.676
Degrees of Freedom: 2-1 = 1
Probability Value (p-value) : 0.50 < p < 0.30
This p-value reflects that there is no significant difference between our observed values and the expected values in this experiment.
Male Tan Body (Wild Type) x Female Ebony Body Cross: (F1 Generation)
Number of Ebony Males: 0
Number of Ebony Females: 1
Number of Tan Males: 11
Number of Tan Females: 8
Total Number of Flies: 20
2 Integers = 20/2 = 10 (Expected Number)
Formula: X 2 = (Observed – Expected) 2 / Expected
Tan Males: X 2 = (11-10) 2 /10 = 0.10
Tan Females: X 2 = (8-10) 2 /10 = 0.40
Ebony data : 0
Total Chi Square : 0.10+0.40 = 0.50
Degrees of Freedom: 2-1 = 1
Probability (p-value) : 0.50 < p < 0.30
This p-value reflects that there is no significant difference between our observed values and the expected values in this experiment.
Male Tan Body (Wild Type) x Female Ebony Body Cross: (F2 Generation)
Number of Ebony Males: 11
Number of Ebony Females: 8
Number of Tan Males: 11
Number of Tan Females: 15
Total Number of Flies: 45
Expected Numbers:
33.75: Tan flies
11.25: Ebony flies
Formula: X 2 = (Observed – Expected) 2 / Expected
Ebony Flies: X 2 = (19-11.25) 2 /11.25 = 4.3
Tan Flies: X 2 = (26-33.75) 2 /33.75 = 1.77
Total Chi Square : 1.77+4.3 = 6.07
Degrees of Freedom: 2-1 = 1
Probability Value (p-value) : 0.05< p < 0.01
There is a significant difference between observed and expected values.
Discussion
The expected phenotype ratios and the observed data for both crosses did not match the observed experimental data. This may be because the original true breed’s offspring might have sexually reproduced before they could have been counted. The offspring from the true breed were not all heterozygous dominant. There were some ebony bodies present in the count.
There is a significant difference between observed and expected values in the Chi Square Analysis in both crosses as well. This might be because our sample size was small. Therefore, we did not have enough data to be accurate. Our observed data may not be efficient.
Since the F2 generation was not successful for the female tan body and male ebony body cross, there is no data for it and no conclusion can be made from the F2 generation.
The F2 generation for the female ebony body and the male tan body cross was successful. However, there was a significant difference in the observed and expected values for the Chi Square Analysis. The F2 generation’s sample size was relatively small so there was not enough data to be accurate.
Since the fruit flies were only counted once a week during class, there was room for error. To reduce the room for error in the future, the fruit flies should be constantly observed because of how fast their life cycles are. If more time was provided, I would redo the experiment and make sure to observe the fruit flies constantly. Since the experiment was not fully successful, there are still questions about inheritance. If I were to redo the experiment successfully, I would be able to formulate questions to perform a follow-up experiment.
There is a significant difference between observed and expected values in the Chi Square Analysis in both crosses as well. This might be because our sample size was small. Therefore, we did not have enough data to be accurate. Our observed data may not be efficient.
Since the F2 generation was not successful for the female tan body and male ebony body cross, there is no data for it and no conclusion can be made from the F2 generation.
The F2 generation for the female ebony body and the male tan body cross was successful. However, there was a significant difference in the observed and expected values for the Chi Square Analysis. The F2 generation’s sample size was relatively small so there was not enough data to be accurate.
Since the fruit flies were only counted once a week during class, there was room for error. To reduce the room for error in the future, the fruit flies should be constantly observed because of how fast their life cycles are. If more time was provided, I would redo the experiment and make sure to observe the fruit flies constantly. Since the experiment was not fully successful, there are still questions about inheritance. If I were to redo the experiment successfully, I would be able to formulate questions to perform a follow-up experiment.